About
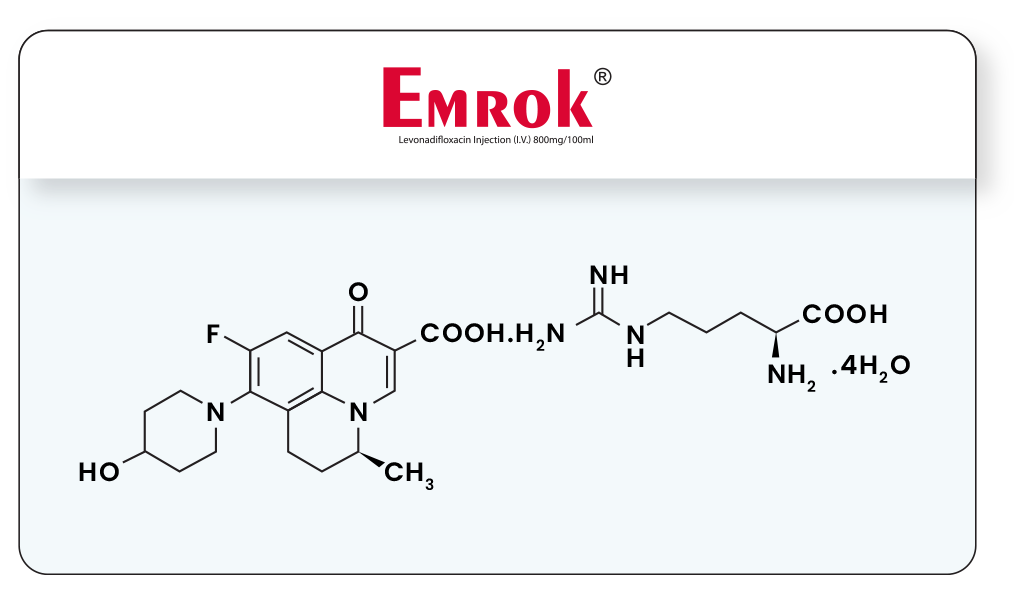
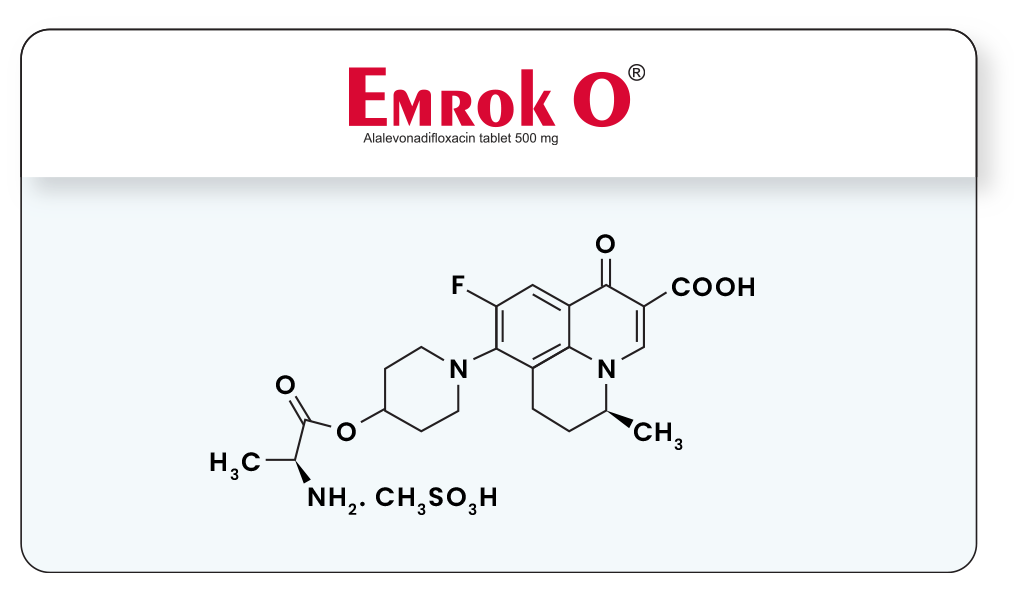
EMROK is an intravenous formulation of arginine salt of Levonadifloxacin equivalent to 800 mg of Levonadifloxacin. EMROK O is an oral formulation of mesylate salt of Alalevonadifloxacin (prodrug of Levonadifloxacin) equivalent to 500 mg of Alalevonadifloxacin.
Discovery
MRSA remains one of the leading infectious causes of mortality in hospitalized patients worldwide. The scenario was more acute in early 2000s when vancomycin was the mainstay of therapy for MRSA infections. Introduction of linezolid though brought an important alternative option, unmet need for an anti-MRSA antibiotic with safety for longer duration use, bactericidal activity, tissue penetration and option for intravenous to oral switch continued to exist. To address this unmet need, fluoroquinolone class was an attractive choice since the class is bestowed with potent bactericidal features, good tissue distribution including in lungs and oral bio-availability. However, the challenges were achieving anti-MRSA activity, low-propensity for resistance selection and safety. Yet, Wockhardt ventured in to breaking these challenges by working on compounds with 8-methoxy and 8-methyl core and could obtain compounds (WCK 1152 and WCK 1654) with modest improvement in MRSA activity compared to moxifloxacin.
At the same time, in parallel, elaborate literature survey had led to some Japanese publications that reported interesting anti-MRSA activity for (±)-nadifloxacin (ingredient of topical anti-acne product). It appeared that benzoquinolizine core and a distinctive 4-hydroxy-piperidine side chain in the structure was responsible for anti-MRSA activity. At that time, synthesizing the racemic nadifloxacin was a difficult task. But, the medicinal chemistry team was able to synthesize the (±)-nadifloxacin and further synthesized even the chiral-pure 'R’ and ‘S’ isomers of nadifloxacin. On separating ‘R’ and ‘S’ isomers of nadifloxacin, some interesting biological findings have emerged. These were all related to ‘S’ isomer which had good oral bioavailability and intravenous tolerability in rodents and importantly, improved anti-MRSA activity. The latter was the result of separation from R isomer which had only weak anti-MRSA activity.
Moreover, ‘S’ isomer was not a substrate of Nor A efflux pump which is ubiquitous in S. aureus. The mechanism-of-action studies conducted at Dr. David Hooper’s lab in Harvard Medical School revealed that S-(-)-nadifloxacin had significantly superior inhibition of staphylococcal DNA gyrase compared to older fluoroquinolones such as ciprofloxacin. Later, extensive studies conducted at Wockhardt showed that S-(-)-nadifloxacin exhibited low-resistance selection potential in staphylococci.
Our initial aim of developing intravenous anti-MRSA drug employing S-(-)-nadifloxacin met with another hurdle as it caused phlebitis after repeated administration in toxicity studies. This was resolved after many months of efforts culminating in development of L-arginine salt of (S)-Nadifloxacin (Levonadifloxacin) which has been termed as WCK 771 (Levonadifloxacin). Although arginine salt satisfied the need for an IV anti-MRSA drug, its oral bioavailability was not adequate. Subsequently, >50 pro-drugs had been studied and among them mesylate salt of L-alanine ester of Levonadifloxacin, termed as WCK 2349 (Alalevonadifloxacin) was identified. This prodrug had high solubility (200mg/mL) across the alimentary tract pH range of 2 – 9, gastric stability, rapid, total absorption followed by esterase mediated cleavage providing plasma Levonadifloxacin concentration profile superimposable with IV formulation of Levonadifloxacin.
Molecular structures of EMROK & EMROK O
References: 1. Bhagwat, S.S., et al., Levonadifloxacin, a Novel Broad-Spectrum Anti-MRSA Benzoquinolizine Quinolone Agent: Review of Current Evidence. Drug Des Devel Ther, 2019. 13: p. 4351-4365.
2. de Souza N.J., et al. A chiral benzoquinolizine-2-carboxylic acid arginine salt active against vancomycin-resistant
Staphylococcus aureus. J Med Chem. 2005 Aug 11;48(16):5232-42.
Indication and Uses
DOSAGE FORM AND STRENGTH
Intravenous Injection (Single use). Emrok is a clear, colourless to pale yellow, sterile, non-pyrogenic, 100 ml isotonic solution intended for intravenous infusion.
THERAPEUTIC INDICATION
Emrok is indicated in adults (≥ 18 years of age) for the treatment of Acute Bacterial Skin and Skin Structure Infections (ABSSSI) including diabetic foot infections and concurrent bacteraemia caused by susceptible isolates of the following:
Gram-positive organisms
Staphylococcus aureus (methicillin-resistant, methicillin-susceptible, quinolone-resistant, quinolone-susceptible isolates), Streptococcus pyogenes, Enterococcus faecalis, Streptococcus dysgalactiae ssp. dysgalactiae, Streptococcus agalactiae. It is critical that a Gram-negative therapy is initiated if a concomitant Gram-negative infection is suspected or documented.
Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Emrok and other antibacterial drugs, Emrok should be used only to treat infections that are proven or strongly suspected to be caused by the susceptible strains of above listed bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
POSOLOGY
For the treatment of adults (≥ 18 years of age) with ABSSSI including diabetic foot infections, and concurrent bacteraemia; the recommended dosage regimen is as follows
- Administer 800 mg every 12 hours by intravenous infusion over a period of 90 minutes for 7-14 days or,
- Following appropriate duration of intravenous therapy, based on physician discretion, switch over to oral Alalevonadifloxacin mesylate (Emrok O) 1000 mg (two tablets of 500 mg each) every 12 hours. Emrok O tablets to be swallowed sequentially with sufficient amount of water and may be taken independent of food.
Note: For improved injection site tolerability, Emrok should be administered by intravenous infusion over a period of 90 minutes. Rapid or bolus intravenous injection or infusion of less than 90 minutes must be avoided. Emrok is not intended for intra-arterial, intramuscular, intrathecal, intraperitoneal, or subcutaneous administration.
Hepatic impairment
No dosage adjustment is required in patients with hepatic impairment (See “USE IN SPECIAL POPULATIONS” for more details)
Renal impairment
Pharmacokinetic studies with Emrok in renal impaired patients have not been conducted.
Method of Administration
Emrok is supplied in a single, ready-to-use glass bottle. The Emrok bottle should be inspected visually for particulate matter and discolouration prior to administration. Levonadifloxacin may exhibit a yellow colour that can intensify over time without adversely affecting potency. Since the bottles are for single-dose only, any unused portion remaining in the bottles should be discarded.
Emrok should be administered every 12 hours over 90 minutes by direct intravenous infusion or through a Y-type intravenous infusion set, which may already be in place. Since no data is available on the compatibility of Emrok intravenous injection with other intravenous drugs, additives or other medications should not be added to Emrok intravenous solution or infused simultaneously through the same intravenous line. If a common intravenous line is being used to administer other drugs in addition to Emrok, the line should be flushed before and after each Emrok infusion with 0.9% Sodium Chloride for Injection.
Important safety Information
CONTRAINDICATIONS
- In individuals with a known hypersensitivity to Levonadifloxacin or other quinolone antibacterials, or to any of the excipients.
- In patients with a history of tendon disorders
- In children or growing adolescents (<18 years of age)
- During pregnancy and lactation
Tendinitis and Tendon Rupture
Fluoroquinolones, including Emrok, are associated with an increased risk of tendinitis and tendon rupture in all ages and can occur within hours or weeks of starting fluoroquinolone therapy, or as long as several months after completion of fluoroquinolone therapy. This risk of developing fluoroquinolone-associated tendinitis and tendon rupture is increased in patients above 60 years of age, in patients taking corticosteroid drugs, and in patients with kidney, heart, and/or lung transplant. In the Phase III clinical study, there was no occurrence of tendinitis or tendon rupture reported in the patients treated with Emrok. At the first sign of tendon pain, swelling, or inflammation, discontinue Emrok injection, avoid exercise and use of the affected area, and inform promptly to a healthcare provider. Avoid Emrok in patients who have a history of tendon disorders or have experienced tendinitis or tendon rupture.
Peripheral Neuropathy
Fluoroquinolones have been associated with an increased risk of peripheral neuropathy. Symptoms may occur soon after initiation of fluoroquinolones and may be irreversible in some patients. In the Phase III clinical study, there was no occurrence of peripheral neuropathy reported in the patients treated with Emrok. In order to minimize the development of an irreversible condition, Emrok should be discontinued immediately if the patient experiences symptoms of peripheral neuropathy including pain, burning, tingling, numbness, and/or weakness or other alterations of sensation including light touch, pain, temperature, position sense, and vibratory sensation and/or motor strength. Avoid fluoroquinolones, including Emrok in patients who have previously experienced peripheral neuropathy.
Central Nervous System Effects
Fluoroquinolones are associated with an increased risk of central nervous system (CNS) reactions, including: convulsions, increased intracranial pressure (including pseudotumor cerebri), disturbances in attention, disorientation and toxic psychosis. Fluoroquinolones may also cause CNS reactions of nervousness, agitation, insomnia, anxiety, nightmares, paranoia, dizziness, confusion, tremors, hallucinations, depression and suicidal thoughts or acts, memory impairment, serious disturbances in mental abilities called delirium.
The mental health side effects are more prominent and more consistent across the systemic fluoroquinolone drug class. These adverse reactions may occur following the first dose. In the Phase III clinical study, there was no occurrence of any of the above mentioned drug-related reactions reported in the patients treated with Emrok. If these reactions occur in patients receiving Emrok, discontinue Emrok immediately and institute appropriate measures. As with all fluoroquinolones, use Emrok when the benefits of treatment exceed the risks in patients with known or suspected CNS disorders (e.g. severe cerebral arteriosclerosis, epilepsy) or in the presence of other risk factors that may predispose to seizures or lower the seizure threshold.
Exacerbation of Myasthenia Gravis
Fluoroquinolones have neuromuscular blocking potential and may exacerbate muscle weakness in persons with myasthenia gravis. Post-marketing serious adverse reactions, including death and requirement for ventilator support, have been associated with fluoroquinolone use in persons with myasthenia gravis. In the Phase III clinical study, no patients with myasthenia gravis were enrolled. Avoid Emrok in patients with known history of myasthenia gravis.
Hypersensitivity Reactions
Serious and occasionally fatal hypersensitive (anaphylactic) reactions, some following the first dose, have been reported in patients receiving fluoroquinolone therapy. Levonadifloxacin did not induce any hypersensitive reaction in a Guinea pig maximization test. In case of serious anaphylactic reactions, institute immediate emergency treatment with epinephrine and other resuscitative measures, including oxygen, intravenous fluids, antihistamines, corticosteroids, pressor amines, and airway management, as clinically indicated. In the Phase III clinical study there was no occurrence of a hypersensitivity reaction reported in patients treated with Emrok. Emrok should be discontinued immediately at the first appearance of a skin rash or any other sign of hypersensitivity.
Photosensitivity/Phototoxicity
Quinolones have been shown to cause photosensitivity reactions to ultraviolet (UVA and UVB) and visible radiation in patients. However, preclinical studies in Swiss mice have shown that Levonadifloxacin has a lower risk to induce photosensitivity (UVA) compared to Sparfloxacin (positive control). Moderate to severe photosensitivity/phototoxicity reactions (e.g., burning, erythema, exudation, vesicles, blistering, oedema), can be associated with the use of fluoroquinolones after sun or UV light exposure. Therefore, excessive exposure to these sources of light should be avoided. Drug therapy should be discontinued if phototoxicity occurs. In the Phase III clinical study, there was no occurrence of a photosensitivity reaction reported in patients treated with Emrok.
Clostridium difficile-Associated Diarrhoea
Clostridium difficile-associated diarrhoea (CDAD) has not been reported in any of the clinical trials of Emrok. Diarrhoea, particularly if severe, persistent and/or bloody, during or after treatment with Emrok (including several weeks after treatment), may be symptomatic of CDAD. CDAD may range in severity from mild to life threatening, the most severe form of which is pseudomembranous colitis. If CDAD is suspected or confirmed, Emrok must be stopped immediately and appropriate treatment should be initiated without delay (e.g. oral metronidazole or vancomycin). Appropriate infection control measures should be undertaken to reduce the risk of transmission. Medicinal products inhibiting the peristalsis are contraindicated in this clinical situation
Dysglycaemia
Disturbances in blood glucose, including both hypoglycaemia and hyperglycaemia have been reported with fluoroquinolones, usually in diabetic patients receiving concomitant treatment with an oral hypoglycaemic agent (for example sulphonylurea) or with insulin. In diabetic patients, careful monitoring of blood glucose is recommended. Low blood sugar levels, also called hypoglycaemia, can lead to coma. In the Phase III clinical study, there was no occurrence of hypoglycaemia and hyperglycaemia reported with Emrok O and three cases of hyperglycaemia/increased blood glucose reported with Emrok were considered to be unrelated to the study medication. If a hypoglycaemic reaction occurs, Emrok should be discontinued and appropriate therapy should be initiated immediately.
Prolongation of QT interval
In a randomized, positive-and placebo-controlled, thorough QT/QTc study conducted in US, 48 healthy subjects received Emrok O supratherapeutic dose (2600 mg), oral moxifloxacin (400 mg) and placebo. Emrok O at the supratherapeutic dose (2600 mg) did not cause any clinically significant changes on the electrocardiogram including the QTc interval.
Development of Drug-Resistant Bacteria
Prescribing Emrok in the absence of a proven or strongly suspected bacterial infections or prophylactic indication is unlikely to provide benefit to the patient and could increase the risk of the development of drug resistant bacteria.



